Supercharging electrochemistry
with aerogel alloys
What makes a good catalyst?
Catalysts lower the activation energy for electrochemical reactions that are fundamental to water-splitting, fuel cells, batteries, and CO₂ conversion. Better catalysts mean less energy is required to make the desired products. Early variants of M1’s aerogel alloy catalysts have outperformed commercial benchmarks in the key performance criteria of water-splitting electrolysis: cost, catalytic efficiency and durability. This means plant operators can reduce their energy bills and extend the operational life of their equipment.
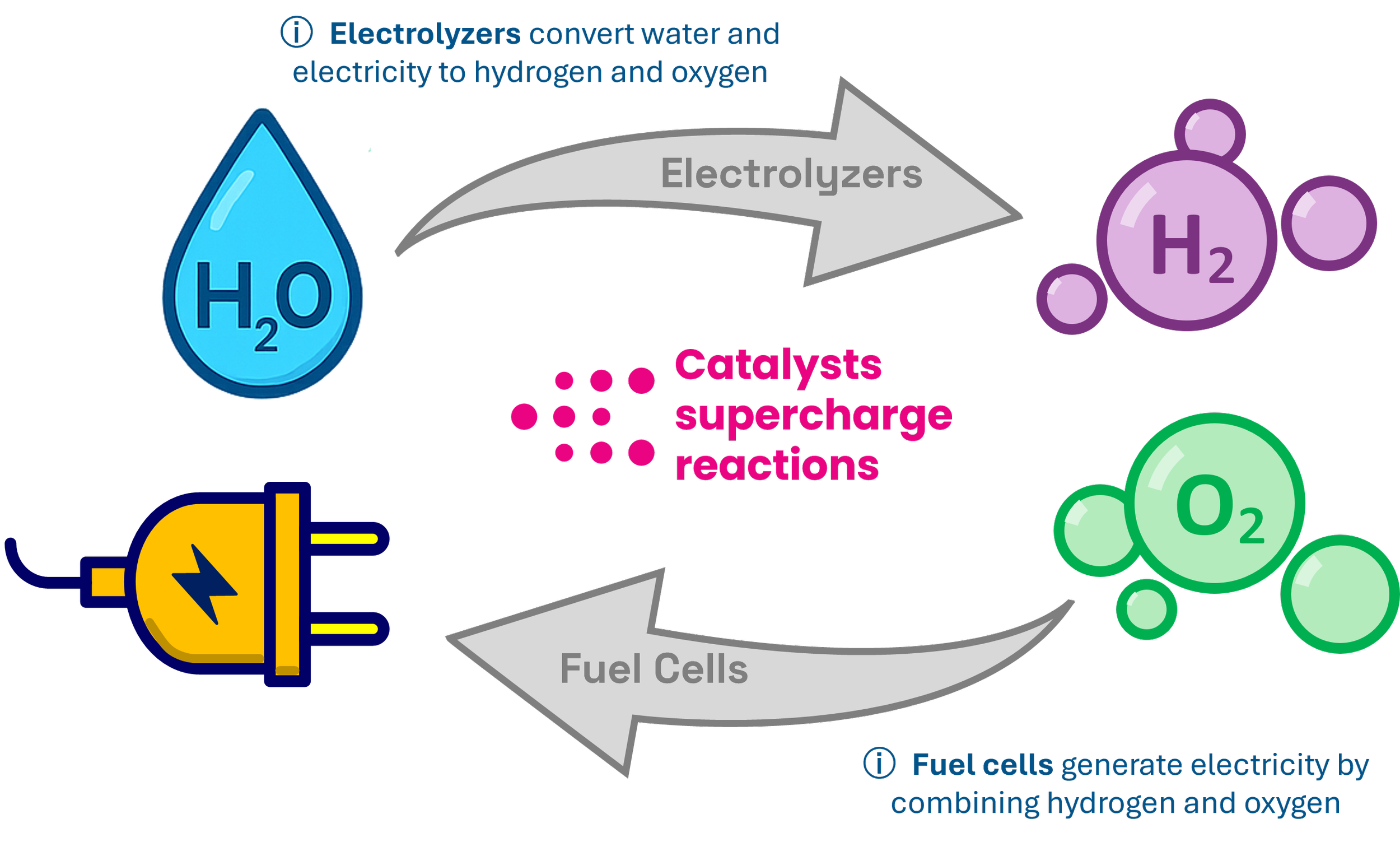
Why aerogel alloys?
M1 Catalysts selected aerogels for their highly-tunable properties, amorphous three dimensional structure, and unmatched porosity which maximizes the number of active sites available for catalytic reactions. Interconnected pores facilitate rapid mass transport of reactants and products, reducing diffusion limitations and boosting reaction efficiency. The aerogel alloy structure exhibits mechanical flexibility that withstands lattice strain and contributes to exceptional durability in harsh operating conditions.

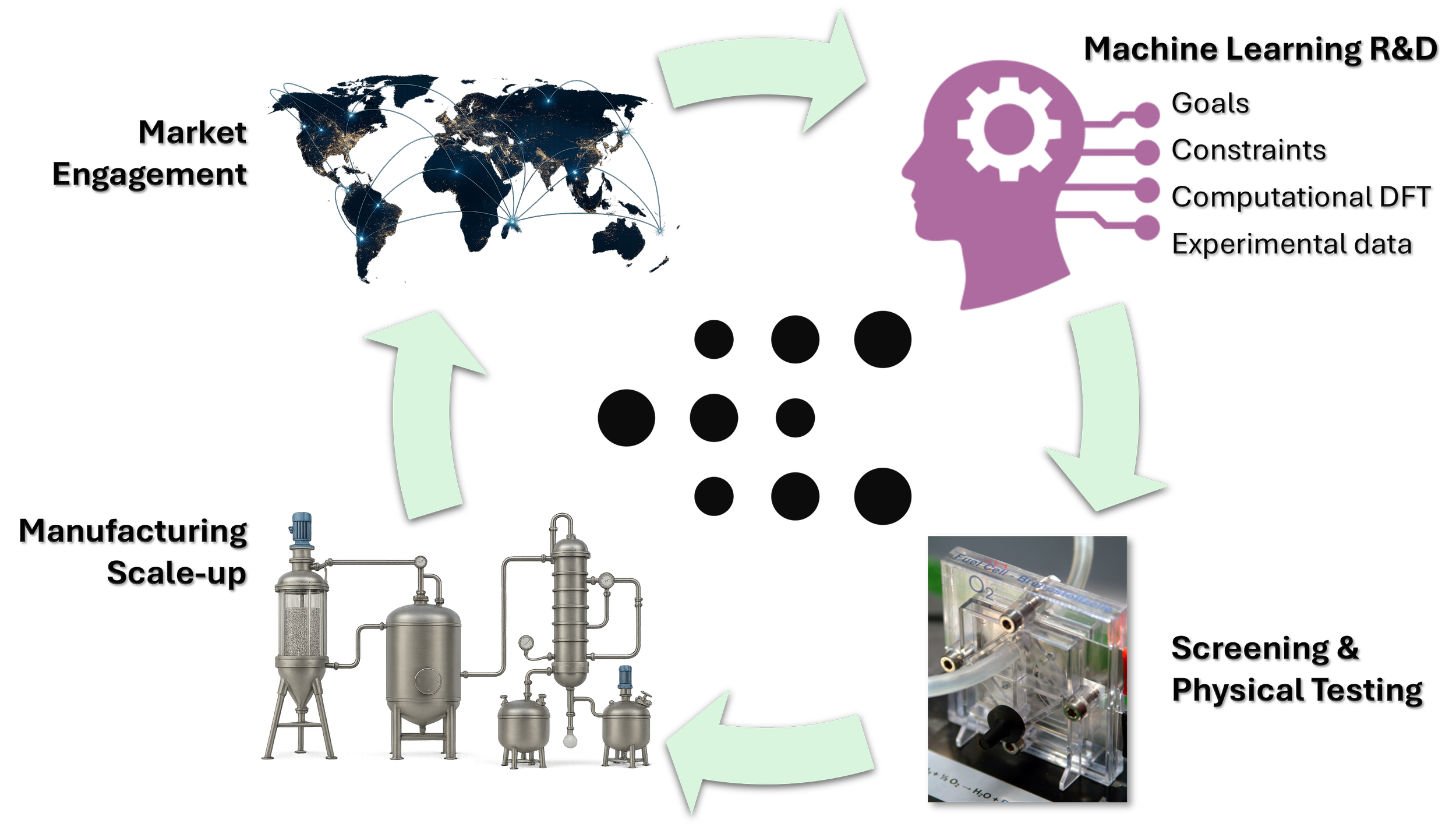
Value creation at M1
Value is determined by the market, so M1 Catalysts uses the market both as a driver for R&D activities and the measuring stick for commercial offerings. By integrating state-of-the-art machine learning R&D with advanced testing and manufacturing methods, M1 accelerates the delivery of useful and cost-effective catalyst products that can meet the evolving energy needs of society.

Early promise for AEM electrolysis
AEM electrolysis is the fastest-growing electrolyzer segment, combining PEM’s efficiency with alkaline’s affordability and enabling operation with pure water and intermittent renewable power—ideal for decentralized clean hydrogen production. Market forecasts show enormous growth potential as durability challenges are solved.
Catalyst aerogel alloy powder with excellent durability for OER.
[PATENTS PENDING]
Catalyst aerogel alloy powder with excellent durability for HER.
[PATENTS PENDING]
RDE testing of M1’s prototype aerogel catalysts show improvements of 1.19-1.36x catalytic activity and 6x durability compared with industry benchmarks.
Contact us to request technical information, samples, or inquire about pilot opportunities.

Foundational research
M1 Catalysts leverages the discoveries of Dr. Yuehe Lin of Washington State University (WSU), one of the world’s leading scientists in electrochemistry, nanotechnology, materials science, and biosensors. Since 2016, Dr. Lin has published numerous journal papers and reviews in his focus area of aerogel electrocatalysts. M1 Catalysts is commercializing Dr. Lin’s most exciting recent discoveries and expanding on the core intellectual property licensed exclusively from WSU. If you’re ready to dive into the science, we hope you’ll appreciate this curated list of Dr. Lin’s publications.

Li, D., Park, E. J., Zhu, W., Shi, Q., Zhou, Y., Tian, H., Lin, Y., Serov, A., Zulevi, B., Baca, E. D., Fujimoto, C., Chung, H. T., & Kim, Y. S. (2020). Highly quaternized polystyrene ionomers for high performance anion exchange membrane water electrolysers. Nature Energy, 5(5), 378–385. https://doi.org/10.1038/s41560-020-0577-x
Zhu, C., Fu, S., Song, J., Engelhard, M. H., Li, X., Du, D., & Lin, Y. (2019). Nickel–Iron Disordered Nanofoam Catalysts for Electrochemical Water Splitting. In Y. Lin & S. Fu (Eds.), Electrochemical Water Splitting: Materials, Processes and Architectures (pp. 183–210). Wiley-VCH. https://doi.org/10.1002/9783527843565.ch7
Fu, S., Song, J., Zhu, C., Xu, G.-L., Amine, K., Sun, C.-J., Li, X., Engelhard, M. H., Du, D., & Lin, Y. (2018). Ultrafine and highly disordered Ni₂Fe₁ nanofoams enabled highly efficient oxygen evolution reaction in alkaline electrolyte. Nano Energy, 44, 319–326. https://doi.org/10.1016/j.nanoen.2017.12.010
Zhu, C., Fu, S., Song, J., Engelhard, M. H., Li, X., Du, D., & Lin, Y. (2018). Disordered NiFe2O4 nanofoam as an efficient electrocatalyst for oxygen evolution reaction. Materials Horizons, 5(2), 151–158. https://doi.org/10.1039/C8MH01397G
Zhu, C., Fu, S., Song, J., Engelhard, M. H., Li, X., Du, D., & Lin, Y. (2017). Nickel–Iron Disordered Nanofoam Catalysts for Electrochemical Water Splitting. Advanced Energy Materials, 7(1), 1601555. https://doi.org/10.1002/aenm.201601555
Fu, S., Zhu, C., Song, J., Engelhard, M. H., Li, X., Du, D., & Lin, Y. (2016). Nickel–Iron Disordered Nanofoam Catalysts for Electrochemical Water Splitting. ACS Energy Letters, 1(5), 792–796. https://doi.org/10.1021/acsenergylett.6b00408
Zhu, C., Fu, S., Song, J., Engelhard, M. H., Li, X., Du, D., & Lin, Y. (2016). Efficient Synthesis of MCu (M = Pd, Pt, and Au) Aerogels with Accelerated Gelation Kinetics and their High Electrocatalytic Activity. Advanced Materials, 28(39), 8779–8783. https://doi.org/10.1002/adma.201602546
Zhu, C., Li, H., Fu, S., Du, D., & Lin, Y. (2016). Highly efficient nonprecious metal catalysts towards oxygen reduction reaction based on three-dimensional porous carbon nanostructures. Chemical Society Reviews, 45(15), 517–531. https://doi.org/10.1039/C5CS00670H


